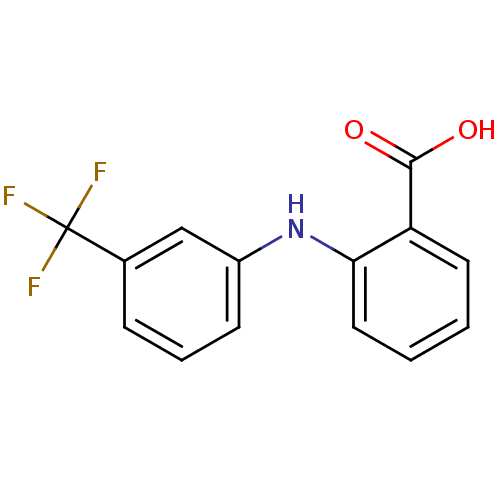
BDBM17636 2-{[3-(trifluoromethyl)phenyl]amino}benzoic acid::Arlef::CHEMBL23588::Flufenamic acid::Nichisedan::US20240002326, Compound Flufenamic acid::US9271961, Flufenamic Acid
SMILES OC(=O)c1ccccc1Nc1cccc(c1)C(F)(F)F
InChI Key InChIKey=LPEPZBJOKDYZAD-UHFFFAOYSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 4 hits for monomerid = 17636
Found 4 hits for monomerid = 17636
Affinity DataIC50: 16nMAssay Description:Compounds may be evaluated as selective reversible inhibitors of AKR1C3 by screening them against homogeneous recombinant AKR1C1-AKR1C4 expressed in ...More data for this Ligand-Target Pair
Affinity DataIC50: 9.30E+3nMAssay Description:Inhibition of COX2 (unknown origin)More data for this Ligand-Target Pair
Affinity DataIC50: 20nMAssay Description:Inhibition of recombinant COX2More data for this Ligand-Target Pair
Affinity DataIC50: 16nMAssay Description:Inhibition of COX2 expressed in baculovirus infected SF-21 cells assessed as formation of PGH2 from PGG2 using arachidonic acid as substrate preincub...More data for this Ligand-Target Pair
 3D Structure (crystal)
3D Structure (crystal)